The customer transition from liquid to powder is complicated and several roadblocks must be addressed. The high-temperature-resistant market is no exception. The use of silicone polymers in this market posed an additional set of hurdles, which delayed the conversion from the late 1980s to the early ‘90s. Silicones by nature have a low softening point and are not compatible with organic polymers. The former would limit the quantity of silicone incorporated into an organic system and therefore limit the high temperature performance of the product. The problem with certain silicone resins and their compatibility limited their use and market acceptance. The re-engineering of silicone technology to increase the softening point and use the current composition enables formulators to develop technologies for the next generation.
Background
The use of silicone resins in powder coatings began in the late 1980s, and the technology became commercial in the mid ‘90s (see sidebar). During this time, formulations were developed using conventional silicone technology and were composed of relatively low levels of silicone. Silicone resins increase the high temperature resistance of coatings and, because of this, they were limited to applications with medium- to high-temperature-resistance such as barbecue grills, smokers, and cookware applications. In the late ‘90s, silicone manufacturers focused on developing new technologies that would enable formulators to maximize silicone content in powder coatings. This would improve the performance of current technology and open new markets to the possibility of using powder. Some of the new applications include coatings for lighting fixtures (whites), appliances (ovens, stoves, etc.), muffler and exhaust systems. During the last five years, the majority of the high-temperature-resistant powder coatings market has been supplied by U.S. manufacturers, with very little supplied by European manufacturers.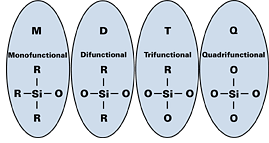
Review
Silicone manufacturers describe different polymers by the types of monomers used during synthesis. Silicon is a tetravalent atom, which forms four sigma bonds in its ground state, and is considered reactive through the oxygen bond (see Figure 1). If one oxygen is bonded to the silicon atom, it is referred to as a monofunctional, or “M” unit; silanes containing two silicon-oxygen bonds are difunctional, or “D” units. “T” units are trifunctional monomers, and “Q” represents a quadrifunctional silane. In addition, silicon has a high affinity for oxygen and will form a high energy Si-OSi bond (108–110Kcal/mole), whereas the bond energy of the C-CC is 82–85 Kcal/mole and the C-OC bond energy is 84–87 Kcal/mole. Therefore, resins used in powder coatings are mainly composed of “T” units, which maximize the number of Si-O bonds. There may be some “D” units present to increase flexibility. Since the Si-OSi bond energy is approximately 20% higher than its organic counter part, the incorporation of a high degree of Si-O (siloxy) units will increase the heat- and UV resistance of the system.


Incorporation
The modification of coatings with silicone polymers can be achieved by either pre-reacting or post blending the silicone with organic polymers. There are four basic types of reactions that occur with silicone resins: alkoxy/water, alkoxy/hydroxyl, silanol/silanol and silanol/hydroxyl. The pre-reaction method is normally carried out by a resin manufacturer, and involves reacting an alkoxy or silanol functional resin with a hydroxyl functional organic resin (see Figure 2). This reaction requires heat and, if the silicone is alkoxy functional, will require distillation of an alcohol. However, the blending method uses conventional equipment to mix the silicone resin and other coating raw materials together.
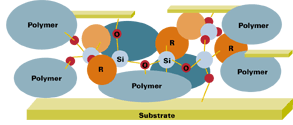
Film Formation
Film formation begins with coating application, which can be done by either tribo or corona spray. After application, the coating passes through an oven set to cure at ~220°C (420°F). The curing process will begin with polymers going through a melt phase, enabling the reactive groups on the polymer to become mobile with enough energy to crosslink (see Figure 4). The silanol groups on the silicone will crosslink with other silanol groups (homopolymerize) to form the Si-O-Si matrix. The silanol groups will also react with hydroxyl groups on neighboring organic polymers to form a thermoset system. This is in addition to the reactions occurring with the organic binder (TGIC/polyester, GMA acrylic/phenol, epoxy/polyester or epoxy/cresol/novolac, etc.).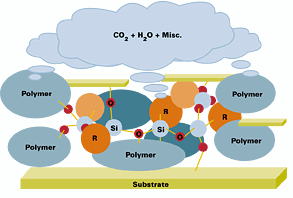
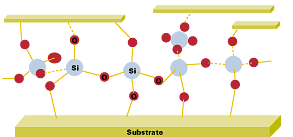
New Product Developments
The next generation of powder coatings will push the envelope on high temperature performance by maximizing the level of silicone resin. Complete oxidation of the film will leave an inorganic skeleton composed of the oxidized silicone resin and inorganic pigments, as illustrated in Figure 6.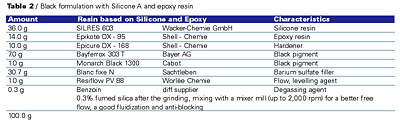

Applications
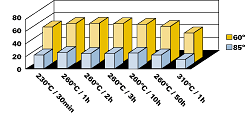
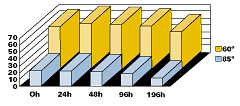
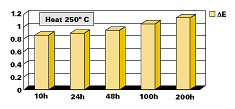
All results are based on powders, which were applied at a 20–40 (Greek Mu) (0.8–1.6mils) film thickness. The powder particle size was from 10 to 60 (Greek Mu). The coatings were applied using a Walther Pilot spray system (Tribo) and cured at 220–250°C (428-480°F) for 30 minutes. Films with higher film thickness will crack and delaminate after exposure to high temperatures. Coatings with high film thickness will also tend to form bubbles from trapped gases (water). The addition of Benzoin or Oxymelt A4 will aid in degassing of these coatings.Powder Preparation
The raw materials were mixed in a Mixaco mixer for 3 minutes at 800 rpm and 20°C (68°F). The mixture was extruded through a single screw extruder (BUSS) with the first zone set at 60°C (140°F) and the second zone set at 100°C (212°F). The torque was 40% and the screw speed was set at 150 rpm. The extrudate was ground into a powder using a Hosokawa high-speed mill set at 12,000 rpm. Fumed silica was added with a setting of 1,400 rpm. The fumed silica helps to increase free flow and fluidization, as well as decrease the tendency of the powder to sinter.
Conclusion
The increase in regulations set by the EPA has forced coatings manufacturers to focus new product developments on powder coatings. Silicone manufacturers have, in turn, focused on developing a new generation of products, which will enable formulators to develop high-performance products. The next generation of products consist of a high level of phenyl or methylphenyl substituents and have a high softening point. The high phenyl content increases organic compatibility and heat resistance. Increasing the softening point will make it possible to formulate powder coatings with high levels of silicone. These new formulations will have excellent gloss stability and non-yellowing characteristics. The modification of organic systems with silicone will also increase UV resistance. These added benefits will make it possible to convert new markets to powder and improve the performance of existing markets.
Sidebar: History of Silicone Resins in Powder Coatings
1994 - First trials with silicone resins in the U.S.1996 - First sale of different silicone resins at the BBQ market
1997 - Silicone resins are established in the heat resistant powder market
1998 - Develop new resins with a higher Tg ( 65–85°C )
1999 - Develop a new resin for heat-resistant white coatings



