Copper Phthalocyanines


In the beginning of the 20th century, commercially available blue pigments consisted mainly of Ultramarine blue, Prussian blue, Indigo derivatives and some azo derivatives. Unfortunately none of these had the desired qualities demanded by the colour industry. Most of these pigments had poor chemical resistance, unsatisfactory acid and/or alkali resistance, insufficient light fastness and poor sublimation fastness. The colour chemists in those days were in need of a cheap and durable pigment that could fill the bluish region of the colour space. The discovery of copper phthalocyanine blue provided them with a satisfactory solution to their problems. Phthalocyanine pigments proved to be outstanding organic pigments because of their incredible resistance to chemical attack, good fastness to heat and light, combined with brightness and cleanliness of shade - all the qualities sought by the colour industry.
Serendipities
When we go back to the past and sort out the history of phthalocyanines, we can see three fascinating serendipity stories.
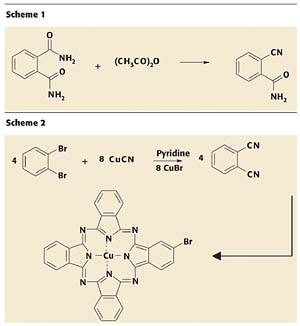
1. In 1907, Braun and Tchermiac tried to synthesize o-cyanobenzamide by dehydrating phthalamide using acetic anhydride (Scheme 1). The scientists failed to get the desired product but instead got a small quantity of a bright bluish compound that was later known as metal-free phthalocyanine. Unfortunately, the scientists failed to recognize the significance of their discovery.
2. It took another 20 years for the second accidental synthesis of the phthalocyanine moiety. Swiss Chemists Von der Weid and de Diesbach attempted to synthesize phthalonitrile from o-dibromobenzene and cuprous cyanide. Surprisingly, the product they isolated from the reaction mass was a deep blue copper complex instead of phthalonitrile (Scheme 2).
Phthalonitrile should be formed here as an intermediate, but it is instantly cyclotetramerizing in the presence of cuprous bromide, a by-product of the reaction, to form semi-bromo copper phthalocyanine.

In 1929, Scottish Dyes obtained a patent for the preparation of phthalocyanine from phthalic anhydride, a metal salt and ammonia. Extensive academic study of the new pigment revealed that the empirical formula was C32H16N8M (where M is metal). In 1934, Sir Reginald Linstead and co-workers deduced the complex macro cyclic structure and coined the name phthalocyanine (phthalo from naphtha (meaning oil) and cyanine meaning blue). M. Robertson confirmed Linstead's structure by using the concept of X-ray crystallographic analysis and showed that the molecule was planar rather than three-dimensional.
Structure
The phthalocyanine system is structurally similar to that of the aza-[18]-annulene series, commonly known as porphine. The two well-known derivatives are haemoglobin, the iron III complex and chlorophyll, the magnesium complex. Chemically one can call copper phthalocyanine the copper complex of tetraazatetrabenzoporphine. The molecule has a completely conjugated structure that exhibits exceptional stability. From the planar structure of the molecule one can see how effectively the metal atom is protected within the interior of the molecule. The central metal atom is covalently bonded with two nitrogens of the porphine ring and also has co-ordinate linkage with two other nitrogens of the same ring. The other factors that contribute to the stability of phthalocyanines are the symmetry of the molecule and the lack of any dipole moment.
A variety of peripheral substitution is possible on a phthalocyanine ring. Numerous copper phthalocyanine analogues have been reported, in which the isoindole unit has been replaced with various other heterocyclic rings. Now it is known that the phthalocyanine macrocycle can complex with cations derived from over 70 different elements. The phthalocyanine complex of Cu2+ ion is so strong that destruction of the macrocycle is required to remove the Cu2+ cation from its central cavity. On the contrary, metal complexes of Li+ and Mg2+ ions are unstable, and these labile ions can be easily removed using dilute acids to produce turquoise blue-coloured metal-free phthalocyanine.
Mechanism of Cyclotetramerization
There are two commercially important processes to produce copper phthalocyanine. One is based on phthalonitrile and the other one uses phthalic anhydride. The phthalonitrile process often yields product with fewer impurities. Another particular advantage of this route is the formation of chloro phthalocyanines when appropriate copper salts are used. Degree of chlorination depends upon the type of copper salt used in the reaction. CuCl2 always gives a product that contains an average of one chlorine atom per copper phthalocyanine molecule, while CuCl can only yield a semi-chloro-copper phthalocyanine. On the other hand, the usage of metallic copper gives plain copper phthalocyanine. Schemes 4-6 describe the various probable reaction mechanisms for the formation of copper phthalocyanines from phthalonitrile.
Reaction of Phthalonitrile and Cupric Chloride
It is suggested that the cyclization is initiated by the attack of chloride anion on an electrophilic carbon of the nitrile group that is activated by the cupric ion. This then triggers the tetramerization, as shown in Scheme 4a.
The chloride nucleophile used for the initiation becomes oxidized to chloronium ion by donating two electrons to the macrocycle complex. The chloronium ion thus formed will either directly undergo aromatic electrophilic substitution at the 4 position, or combine with the other chloride ion to form a chlorine molecule, which in turn takes part in the substitution reaction to form mono chloro phthalocyanine and hydrogen chloride gas. The final product thus formed will contain an average of one chlorine atom per CPC molecule (Scheme 4b).
Reaction of Phthalonitrile and Cuprous Chloride
Here the reaction mechanism may be explained through a free radical pathway where the cuprous ion is oxidized into the cupric state, contributing one electron to the macrocycle formation. The other electron required for the completion of the conjugated porphyrin ring is donated by the chloride nucleophile that is in turn oxidized to chlorine radical (Scheme 5).
In this mechanism, the formation of two copper phthalocyanine rings generates two chlorine radicals that combine together to form a chlorine molecule, which may then react with one of the copper phthalocyanine molecules, as described in Scheme 4. But in this case the final product will have a statistical average of 0.5 chlorine atoms per copper phthalocyanine molecule (semichloro copper phthalocyanine).
Reaction of Phthalonitrile and Copper
When metallic copper is used in the reaction, the assistance of a nucleophile is required to initiate the reaction. Since this nucleophile is regenerated at the end of the reaction, it is needed only in catalytic amounts. Scheme 6 illustrates the formation of copper phthalocyanine (CPC).
CPC - Hydrophobic and Hydrophilic
The CPC molecule has 18 conjugated π electrons that form an electronic cloud on both sides of the molecular plane. This, along with the presence of nitrogen atoms in the porphine ring, contribute to the polar portion of the molecule while the hydrogen atoms of the benzene rings of CPC are highly non-polar. Thus, overall the copper phthalocyanine molecule consists of both hydrophilic and hydrophobic areas. In the crystalline state, each molecule is arranged one above another in order to have the favoured and stable π-π interaction. As a result of this orientation, the majority of the polar faces are buried inside the crystal, and those parts that are exposed to the surface are the nonpolar hydrogen atoms and the substituents of the benzene ring. So in short, a sizeable surface of the phthalocyanine crystal is nonpolar, which contributes to the highly hydrophobic character of the copper phthalocyanine pigments. It is also possible to increase the hydrophilicity of a crystal by altering its shape. R Sappok1 of BASF has done extensive studies on different shaped b CPCs and experimentally demonstrated that isometric pigments are more hydrophilic than the acicular ones, owing to the relatively high concentration of basal planes in the former.Colour
Though the molecular structure of CPC is the key contributor to its colouristic properties, particle shape, size and crystal structure play a decisive role in determining the final shade of the pigment. For example, isometric crystals of b-copper phthalocyanine exhibit greener shade than the acicular ones1. This indicates that one can alter the hue of b-copper phthalocyanine pigment by altering its crystal growth pattern. Some additives or even some impurities (formed during the manufacture of CPC) can promote or inhibit the growth of crystals along certain phases. This will depend upon the polarity and chemistry of the respective phases. A clearer understanding of the nature of a fast-growing phase in a crystal will aid in the design of tailor made additives to control its growth in desired fashion. Similarly, nature and mode of molecular stacking in a crystal influences the final hue of the pigment. For these reasons different polymorphs of CPC show distinctly different hues.Production
In general, a crude copper phthalocyanine is produced by reacting phthalic anhydride, urea, copper/copper salt and ammonium molybdate (catalyst) in high-boiling solvent such as o-nitrotoluene, trichlorobenzene or alkyl benzenes. The choice of the solvent is very important, as it plays a significant role in the generation of impurities. After the completion of the reaction, the solvent is removed by vacuum distillation and the crude product thus obtained is further purified by treatment with dilute acid and alkali solutions to remove basic and acidic impurities present. The product thus obtained is dried and pulverized for use in the next stages of processing.
Crystal Modifications
Unsubstituted copper phthalocyanine blue exists in different crystal modifications. They are named with the Greek letters in the order of their discovery (a, b, g, d, etc). Among these, a and b modifications have gained considerable commercial importance in industry. The e modification is also gaining popularity because of its pure reddish shade.
The exact molecular arrangement of different polymorphs has been revealed by three-dimensional X-ray diffraction techniques. Now it is known that in almost all modifications, the planar copper phthalocyanine molecules are arranged in a one-dimensional stack. The relative arrangements of these stacks, as well as the stacking overlap of the adjacent molecules, are different in different modifications. We can also see a notable difference in the angle between the stapling axis of the stacks and the axis perpendicular to the molecular plane. This non-uniformity is the main reason behind the difference in shade and stability of various polymorphs of CPC (Figure 1).

Thermodynamic Stability of b Modification
In the molecular stacking of the beta modification, it has been suggested that the copper atom at the centre of each molecule is coordinated with the nitrogen atoms of the adjacent molecules. This forms a distorted octahedron geometry, which is very common and favored in complexes of copper. No such coordination is possible in alpha-type crystals. Another positive aspect of the molecular arrangement in beta is the close association of different molecular stacks. In this case, molecules in one stack are arranged in a face to edge style with the molecules in adjacent stacks (Figure 2).
As a result of this arrangement, the π electrons in one molecule and the peripheral hydrogen atoms of the molecules in adjacent stacks are in close proximity. This contributes to a strong association between various molecular stacks inside a crystal. So the entire crystal lattice of the beta modification is stabilized in these two ways. Any factors that affect these stable arrangements may influence the crystal formation in the beta form. The substitution of peripheral hydrogen by other bulky atoms or groups hinders the face to edge packing in beta. Thus, mono and poly halo phthalocyanines exist in the alpha form instead of in beta.

Preparation of b Modification
The crude pigment that is obtained from synthesis mainly consists of crystals of b modifications, but these are very large in size and do not have any pigmentary value. The process of converting these oversized particles to crystals of optimum size with respect to application properties such as colour strength, opacity/transparency, dispersibility, hue, flocculation and crystallization stability is termed finishing.The first step in the finishing process is the preconditioning of coarsely crystalline or agglomerated crude pigment. Here the oversized particles of the crude pigment are ground in a ball mill. The purpose of the preconditioning is to reduce the primary particle size of the pigment down to the range of 0.01 to 0.05 microns. Though particle size reduction occurs during the preconditioning, strong aggregation of the primary particles is inevitable, which results in the formation of agglomerates of 0.5 to 100 microns. Another process that is taking place during grinding is the conversion of b phase to a. J. R. Fryer2 postulated that due to the high shear employed in the grinding process some molecular stacks of b phase dislocate from their original position, causing a gap in the crystal lattice, which in turn leads to the movement of the adjacent stack towards the vacant space. This creates a new arrangement of molecular stacking, which is similar to the arrangement in a modification. The new phase thus created inside a b crystal acts as a nucleus for the growth of a phase and leads to the gradual conversion of b to a. The schematic representation of the formation of a phase nuclei in a b crystal is given in Figure 3.
The a, b mixture thus produced mainly consists of aggregates of tiny brick-shaped crystals packed mainly in a face-to-face fashion. Incorporation of small amounts of crystallizing solvents like xylene at the ball milling stage is often employed to effect the re-conversion of a to b. Solvent treatment is employed for the conversion of a to b and the subsequent crystal growth. M. McGarvey and R.B.McKay3 studied the nature of microstructure of CPC formed after preconditioning and its influence on the final solvent conditioning. They correlate the actual dispersibility of the finished pigment with the grinding conditions employed at the ball milling stage. The preconditioned pigment, having a weakly coherent aggregate structure, requires less time and/or less powerful solvent for a to b conversion than those with more compact and coherent aggregates. Thus it is essential to re-standardize the final solvent finishing process whenever the parameters at the preconditioning stage are altered. Failure to do so may yield products of inconsistent quality. Finishing of the preconditioned crude can be done either by solvent reflux in a mixture of water and crystallizing solvent or by salt grinding in a kneader in the presence of glycols. In both processes, suitable additives are added at key stages to control crystal growth and also to impart optimum dispersibility to the final pigment. As prolonged reflux and usage of strong solvent often lead to over crystallization, the choice of solvent and reflux time is very critical. Over crystallization of the pigment often hampers its tinctorial strength. Various types of CPC derivatives are used in conjunction with other surface-active agents for the surface modification of the finished pigment. The choice of the derivative, surfactant and the conditions employed at the surface treatment stage are the key factors in determining the performance of the finished product in various application media.
Preparation of a Modification
This grade of CPC can be prepared either by dissolving or swelling crude pigment in sulphuric acid, followed by hydrolysis in water. The dissolution of the highly crystalline pigment is due to the protonation of its nitrogen atoms to form the corresponding sulphates. Depending upon the concentration of the sulphuric acid, one molecule of CPC can take up to eight molecules of sulphuric acid (due to the protonation of eight nitrogen atoms of the ring). The degree of agitation, rate of addition of the dissolved CPC, temperature and surfactant, etc., are the key parameters at the hydrolysis stage, which eventually determine the particle size of the finished pigment.Crystallization of a Modification
Pure alpha CPC pigment undergoes a phase transformation followed by crystal growth when treated with aromatic hydrocarbons or subjected to high temperatures. These conditions normally occur when alpha CPC is used in paints/inks containing such solvents and also while processed for the coloration of plastics at high temperatures. The newly formed phase is found to be in the thermodynamically stable b modification and comparatively in a larger crystalline form. This change will affect the shade and reduce the tinctorial strength. The arrangement of molecules in alpha modification is one of the least stable among the polymorphs of CPC. The CPC pigment has a limited solubility in aromatic solvents and due to this the molecules move continuously from the particle surface of a form into the solvent and from the solvent to the particle surface. This process is in a dynamic equilibrium. The molecules that are coming out of the solution naturally crystallize in the most stable b form, and in order to maintain the equilibrium, more and more molecules dissolve and re-crystallize to effect the complete conversion of a to b. Incorporation of suitable quantities of 4-substituted derivatives, especially the 4-chloro copper phthalocyanine, was found to prevent the phase conversion. But the chloro substitution at the 3 position does not have any effect on stabilization. Aluminium, tin and magnesium phthalocyanines have been found to be very effective stabilizers when used in suitable proportions.Surface Treatment
The surface character of a pigment influences various properties in its final application. Interactive forces between individual crystals, between crystals and binders (resins/dispersants/surfactants), and between crystals and solvents have a decisive role in determining flocculation resistance and rheological properties. During the final stages of finishing, the primary pigment particles have to be aggregated to achieve easy isolation from the preparative medium. As strong coherent aggregation adversely affects the dispersibility of the pigments, adequate steps need to be taken to reduce its strength. Generally this is achieved by coating the pigment surface using suitable additives. This process helps to inactivate the active centers of the pigment that take part in strong aggregation. In addition to this, increasing coating coverage will decrease the pigment-to-pigment contact, which in turn decreases the mechanical strength of an aggregate and hence improve its dispersibility. Appropriately chosen additives can also alter the hydrophilic/hydrophobic character of the pigment surface.Various types of additives, including rosins, fatty amines and pigment derivatives have been used in the surface treatment of phthalocyanine pigments. The use of derivatives of phthalocyanines as a surface-treatment additive is very popular in the industry. Owing to their special affinity towards certain planes of pigment crystals, these copper phthalocyanine compounds adhere more strongly to the surface of the CPC pigment than any other conventional additives. One can custom design the nature of the substituent groups of the pigment derivative depending upon the requirement in the final application media. For example, amine substituted pigment derivatives [CPC-(CH2 -NRR')n] establish hydrogen bonding with the acidic components of the application medium, and hence, derivatives of these types will be very effective wherever acidic binders are used. The molecules of the binder that are attached to the pigment surface in this fashion impart sufficient steric barrier to prevent pigment flocculation. Sufficiently long and solvated substituent R, R' groups also provide steric hindrance by extending towards the liquid phase. In this case, the chemistry of the R, R' groups can be tailored to suit the polarity of the application media.
Other phthalocyanine derivatives such as sulphonic acids, sulphonamides, sulphonium salts are also widely used to make special grades of pigments for specific applications. The synthetic design and the treatment methods employed are crucial to get consistent production of good quality pigments.
Acknowledgments
I sincerely thank Colin Gooch (Technical Director, Resene Paints) and my esteemed colleague Chris Monigatti for their constant encouragement and support to complete this article.Ajith Aravindakshan obtained his Ph.D. in Organic Chemistry from Dyes Division of UDCT, Mumbai, India. He has worked with Ciba Specialty Chemicals India Ltd. (Research Fellow) and Meghmani Organics Ltd; (Manager R&D); and is currently working with Resene Paints, New Zealand, as their Pigment Preparations Chemist. He can be reached at ajith_aravind@yahoo.com.
References
1. Sappok, R. J.O.C.C.A. 1978, 61, 299-308.2. Fryer, J.R. Surface Coatings International, 1997, 9, 421-426.
3. McGarvey, M.; McKay, R.B. Surface Coatings International, 1997, 9, 435-440.
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!





